- Ertugliflozin /metformin
Ertugliflozin /metformin is available in four strengths
- ERTUGLIFLOZIN /METFORMIN 2.5/500 containing 3.24 mg ertugliflozin pyroglutamic acid equivalent to 2.5 mg of ertugliflozin and 500 mg of metformin.
- ERTUGLIFLOZIN /METFORMIN 2.5/1000 containing 3.24 mg ertugliflozin pyroglutamic acid equivalent to 2.5 mg of ertugliflozin and 1000 mg of metformin.
- ERTUGLIFLOZIN /METFORMIN 7.5/500 9 containing .71 mg ertugliflozin pyroglutamic acid equivalent to 7.5 mg of ertugliflozin and 500 mg of metformin.
- ERTUGLIFLOZIN /METFORMIN 7.5/1000 containing 9.71 mg ertugliflozin pyroglutamic acid equivalent to 7.5 mg of ertugliflozin and 1000 mg of metformin.
ERTUGLIFLOZIN /METFORMIN (ertugliflozin and metformin) is indicated as an adjunct to healthy eating and physical activity to improve glycaemic management in adults with type 2 diabetes mellitus when treatment with both ertugliflozin and metformin is appropriate.
For the latest PBS indications for ERTUGLIFLOZIN /METFORMIN please see
https://www.pbs.gov.au/pbs/search?analyse=false&term=ERTUGLIFLOZIN&search-type=medicines

General
ERTUGLIFLOZIN /METFORMIN should be taken twice daily with meals, with gradual dose escalation for those initiating metformin to reduce the gastrointestinal side effects due to metformin.
Starting dose of ERTUGLIFLOZIN /METFORMIN (ertugliflozin and metformin) should be Individualised based on the current regimen:
- If an individual is on metformin, switch to ERTUGLIFLOZIN /METFORMIN tablets containing 2.5 mg
ertugliflozin, with a similar total daily dose of metformin.
- If an individual is already treated with ertugliflozin and metformin, switch to
ERTUGLIFLOZIN /METFORMIN tablets containing the same total daily dose of ertugliflozin and a
similar daily dose of metformin.
- Dosing may be adjusted based on effectiveness and tolerability while not exceedingly the
maximum recommended daily dose of 15 mg ertugliflozin and 2,000 mg metformin.
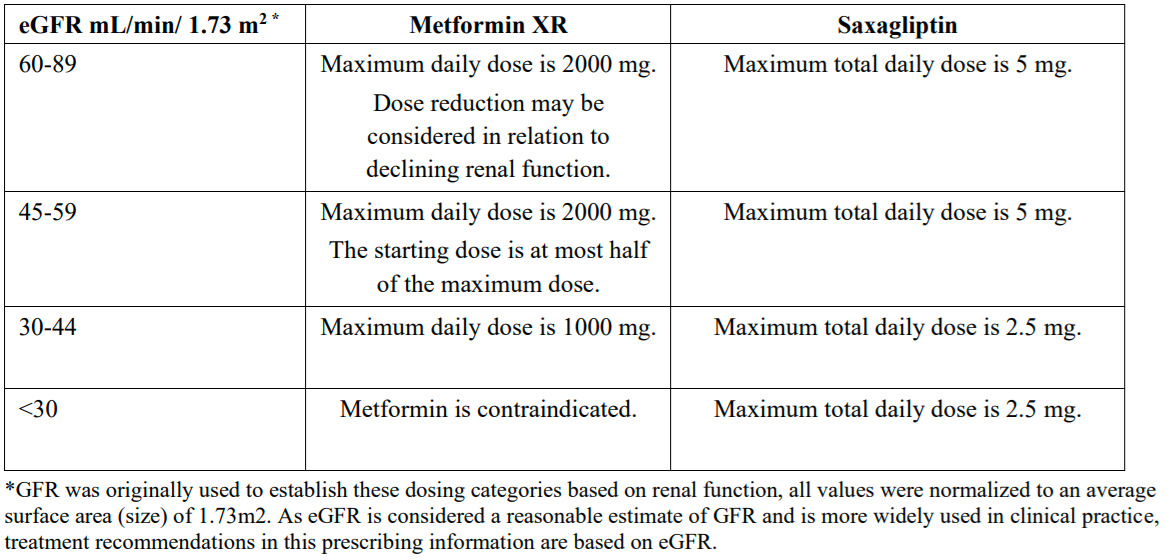
- Renal impairment
An eGFR should be assessed before initiation of treatment with metformin-containing
products and at least annually thereafter. In those at increased risk of further progression of
renal impairment and in the elderly, renal function should be assessed more frequently, e.g.,
every 3 – 6 months.
Initiation of ERTUGLIFLOZIN /METFORMIN is not recommended in those with an eGFR less than
45 mL/min/1.73 m2.
In individuals with an eGFR of 45 to less than 60 mL/min/1.73 m2, individually assess the risk
benefit as HbA1c reduction was only demonstrated in a post hoc analysis for ertugliflozin 15 mg.
Discontinue ERTUGLIFLOZIN /METFORMIN if the person’s eGFR falls persistently below 45 mL/min/1.73 m2.
The maximum daily dose of metformin should preferably be divided into 2 – 3 daily doses.
If no adequate strength of ERTUGLIFLOZIN /METFORMIN is available, individual mono-components should be used instead of the fixed-dose combination.
Discontinuation for iodinated contrast imaging procedures
Discontinue ERTUGLIFLOZIN /METFORMIN at the time of, or prior to, an iodinated contrast imaging procedure in those with an eGFR less than 60 mL/min/1.73 m2; in those with a history of liver disease, alcoholism, or heart failure; or in those who will be administered intraarterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart ERTUGLIFLOZIN /METFORMIN if renal function is acceptable.
- Hepatic impairment
Use of metformin in individuals with hepatic impairment has been associated with some cases of lactic acidosis. ERTUGLIFLOZIN /METFORMIN is not recommended in those with hepatic impairment.
Ertugliflozin and metformin are eliminated in part by the kidney, and therefore, because the elderly are more likely to have decreased renal function, ERTUGLIFLOZIN /METFORMIN should be used with caution as age increases.
- population does this have its own toggle or a bullet point
- Safety and effectiveness of ERTUGLIFLOZIN /METFORMIN in individuals under 18 years of age have not been established.
- History of a serious hypersensitivity reaction to ERTUGLIFLOZIN /METFORMIN, ertugliflozin, metformin, or to any of the excipients
- Those with chronic kidney disease (CKD) receiving dialysis; eGFR <30 mL/min/1.73 m2 (CKD stage 4 and 5). The efficacy of ERTUGLIFLOZIN /METFORMIN is dependent on renal function.
- Acute or chronic metabolic acidosis, including ketoacidosis, with or without coma
- Acute conditions with the potential to alter renal function such as: dehydration, severe infection, shock, or intravascular administration of iodinated contrast agents
- Acute or chronic disease which may cause tissue hypoxia such as: cardiac or respiratory failure, pulmonary embolism, recent myocardial infarction, shock, acute significant blood loss, sepsis, gangrene, pancreatitis.
- During or immediately following surgery where insulin is essential.
- Due to its metformin component, ERTUGLIFLOZIN /METFORMIN is contraindicated in those with conditions that can lead to severe hepatic insufficiency such as: o acute alcohol intoxication o alcoholism
-General
ERTUGLIFLOZIN /METFORMIN should not be used in individuals with type 1 diabetes mellitus.
Metformin
Lactic acidosis-Lactic acidosis is a rare, but serious metabolic complication that can occur due to metformin accumulation during treatment with metformin. When it occurs, it is fatal in approximately 50% of cases. Lactic acidosis is a medical emergency and must be treated in hospital immediately. The risk of lactic acidosis increases with the degree of renal dysfunction. Reported cases of lactic acidosis in individuals on metformin have occurred primarily in those with diabetes with significant renal insufficiency, often in the setting of multiple concomitant medical/surgical problems and multiple concomitant medications. Special caution should be taken in the elderly due to the decrease of renal function with age.
Note: Diabetes Medscheck for referral to CDE for education.
-Surgery
Metformin must be discontinued at the time of surgery under general, spinal, or epidural anaesthesia. Therapy may be restarted no earlier than 48 hours following surgery or resumption of oral nutrition and provided that renal function has been re-evaluated and found to be stable.
Monitoring of renal function
Renal function should be confirmed before initiation of ERTUGLIFLOZIN /METFORMIN therapy, and then at least once a year in those with normal renal function and at least two to four times a year if serum creatinine levels are at or above the upper limit of normal and in elderly individuals.
Decreased renal function in the elderly is frequent and asymptomatic. Special caution should be exercised in situations where renal function may become impaired, for example when initiating antihypertensive or diuretic therapy or when starting treatment with a nonsteroidal anti-inflammatory drug (NSAID).
Note: Diabetes Medscheck for referral to CDE for education.
Alcohol Intake
Alcohol is known to potentiate the effect of metformin on lactate metabolism. Individuals, therefore, should be warned against excessive alcohol intake while receiving ERTUGLIFLOZIN /METFORMIN.
Metformin
-Gastrointestinal disorders
Gastrointestinal symptoms such as nausea, vomiting, diarrhoea, abdominal pain, and loss of appetite are very common (>10%): these occur most frequently during initiation of therapy and resolve spontaneously in most cases. To prevent these gastrointestinal symptoms, it is recommended that this medicinal product be taken in 2 or 3 daily doses. A slow increase of the dose may also improve gastrointestinal tolerability.
-Metabolism and nutrition disorders
-Lactic acidosis is a very rare (<0.01%) but serious metabolic complication that can occur due to metformin accumulation during treatment with metformin. The onset of lactic acidosis is often subtle and accompanied by non-specific symptoms such as malaise, myalgia, respiratory distress, increasing somnolence and non-specific abdominal distress. There may be associated hypothermia, hypotension and resistant bradyarrhythmias with more marked acidosis.
Note: Diabetes Medscheck with referral to appropriate to HCP for education.
-Hepatobiliary disorders
Very rare: liver function test abnormalities or hepatitis requiring treatment discontinuation.
-Skin and subcutaneous tissue disorders
Skin reactions such as erythema, pruritus and urticaria have been reported but the incidence is very rare (<0.01%).
-Nervous system disorders
-Taste disturbance (3 %) is common.
-Vitamin B12 Levels
In controlled, 29-week clinical trials of immediate release metformin, a decrease to subnormal levels of previously normal serum Vitamin B12 levels, without clinical manifestations, was observed in approximately 7% of individuals. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex is, however, very rarely associated with anaemia and appears to be rapidly reversible with discontinuation of metformin or Vitamin B12 supplementation. Measurement of haematologic parameters on an annual basis is advised in those on ERTUGLIFLOZIN /METFORMIN and any apparent abnormalities should be appropriately investigated and managed. Certain individuals (those with inadequate Vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal Vitamin B12 levels). See interventions for further information.
Note: Diabetes Medscheck education and referral to HCP if appropriate.
Ertugliflozin
-Hypotension/Volume depletion
Ertugliflozin, a component of ERTUGLIFLOZIN /METFORMIN, can cause intravascular volume contraction that may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine. Individuals with impaired renal function (eGFR less than 60 mL/min/1.73 m2), the elderly (≥ 65 years), or those on diuretics may be at increased risk for volume depletion or hypotension. Before initiating ERTUGLIFLOZIN /METFORMIN in those with one or more of these characteristics, intravascular volume status should be assessed, and individuals advised on the importance of adequate hydration. Monitor intravascular volume status in addition to blood pressure and renal function after initiating therapy. In case of conditions that may lead to fluid loss (e.g., gastrointestinal illness, heat stress or severe infections), careful monitoring of volume status (e.g., physical examination, blood pressure measurements, laboratory tests including haematocrit) and electrolytes is recommended for anyone receiving ERTUGLIFLOZIN /METFORMIN. Temporary interruption of ERTUGLIFLOZIN /METFORMIN should be considered until the fluid loss is corrected.
Note: Diabetes Medscheck education and referral to HCP if appropriate.
-Ketoacidosis
ERTUGLIFLOZIN /METFORMIN should not be used for the treatment of ketoacidosis. Reports of ketoacidosis, a serious life-threatening condition requiring urgent hospitalisation, have been identified in clinical trials and post marketing surveillance in individuals receiving sodium glucose co-transporter-2 (SGLT2) inhibitors. Fatal cases of ketoacidosis have been reported in those taking SGLT2 inhibitors. ERTUGLIFLOZIN /METFORMIN is not indicated for the treatment of type 1 diabetes mellitus. Individuals treated with ERTUGLIFLOZIN /METFORMIN who present with signs and symptoms consistent with severe metabolic acidosis should be promptly assessed for ketoacidosis regardless of presenting blood glucose levels, as ketoacidosis associated with SGLT2 inhibitors may be present even if blood glucose levels are less than 14 mmol/L. If ketoacidosis is suspected, ERTUGLIFLOZIN /METFORMIN should be discontinued, the person should be evaluated, and prompt treatment should be instituted. Treatment of ketoacidosis generally requires insulin, fluid, potassium, and carbohydrate replacement. Signs and symptoms of ketoacidosis may include excessive thirst, nausea, vomiting, abdominal pain, generalised malaise, and shortness of breath. Restarting SGLT2 inhibitor treatment in those with previous ketoacidosis while on SGLT2 inhibitor treatment is not recommended unless another clear precipitating factor is identified and resolved. Before initiating ERTUGLIFLOZIN /METFORMIN, consider factors in the history that may predispose to ketoacidosis. Factors that predispose a person to ketoacidosis include a low carbohydrate diet, dehydration, acute illness, surgery, previous ketoacidosis, insulin dose reduction, malnourishment / reduced caloric intake or increased insulin requirements due to infections, insulin deficiency from any cause (history of pancreatitis (or pancreatic surgery), and alcohol abuse.
Note: Diabetes Medscheck education and referral to HCP if appropriate.
-Surgery:
ERTUGLIFLOZIN /METFORMIN must be discontinued at least 48 hours prior to major surgery. An increase in other glucose lowering agents may be required during this time. Those scheduled for non-urgent surgery who have not ceased ERTUGLIFLOZIN /METFORMIN should be assessed, and consideration should be given to postponing the procedure. Therapy may be restarted not earlier than 48 hours following surgery, once the person’s condition has stabilised, oral intake is normal, and only if normal renal function has been established, as withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension, and renal impairment.
Note: Diabetes Medscheck education and referral to HCP if appropriate.
-Genital mycotic infections:
Ertugliflozin, a component of ERTUGLIFLOZIN /METFORMIN, increases the risk of genital infections. In trials with SGLT2 inhibitors, those with a history of genital infections and uncircumcised males were more likely to develop genital mycotic infections. Monitor and treat appropriately.
Note: Diabetes Medscheck education and referral to HCP if appropriate.
-Necrotising fasciitis of the perineum:
Post marketing cases of necrotising fasciitis of the perineum (also known as Fournier’s gangrene), a rare, but serious and life-threatening necrotising infection, have been reported in females and males with diabetes mellitus treated with SGLT2 inhibitors. Serious outcomes have included hospitalisation, multiple surgeries, and death. Individuals treated with ERTUGLIFLOZIN /METFORMIN who present with pain or tenderness, erythema, swelling in the genital or perineal area, fever, and malaise should be evaluated for necrotising fasciitis. If suspected, ERTUGLIFLOZIN /METFORMIN should be discontinued, and prompt treatment should be instituted (including broad-spectrum antibiotics and surgical debridement if necessary). Caution is advised in those at increased risk of genital infections including those with recurrent or pre-existing urogenital infections, obesity, immunosuppressed states, smoking, alcohol abuse, end-stage renal or liver failure, and HbA1c >10%.
Note: Diabetes Medscheck education and urgent referral to HCP if appropriate.
- ERTUGLIFLOZIN /METFORMIN has been shown to be bioequivalent to coadministration of corresponding doses of ertugliflozin and metformin tablets.
Ertugliflozin
Following single dose oral administration of 5 mg and 15 mg of ertugliflozin, peak plasma concentrations (median Tmax) of ertugliflozin occur at 1 hour post dose under fasted conditions. Ertugliflozin may be administered with or without food.
Metformin
The absolute bioavailability of a metformin hydrochloride 500 mg tablet given under fasting conditions is approximately 50-60%. Studies using single oral doses of metformin hydrochloride tablets 500 mg to 1,500 mg, and 850 mg to 2,550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination.
Ertugliflozin
Plasma protein binding of ertugliflozin is 93.6% and is independent of ertugliflozin plasma concentrations. Plasma protein binding is not meaningfully altered in those with renal or hepatic impairment.
Metformin is negligibly bound to plasma proteins. At usual clinical doses and dosing schedules of metformin tablets, steady-state plasma concentrations of metformin are reached within 24 48 hours.
Ertugliflozin
Metabolism is the primary clearance mechanism for ertugliflozin. The major metabolic pathway for ertugliflozin is UGT1A9 and UGT2B7-mediated O glucuronidation to two glucuronides. These are present in plasma at levels 2- and 4-times lower than ertugliflozin and are pharmacologically inactive at clinically relevant concentrations.
Metformin Intravenous single-dose studies demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) nor biliary excretion.
Ertugliflozin The mean systemic plasma clearance following an intravenous 100 µg dose was 11.2 L/hr. The mean elimination half-life in those with type 2 diabetes with normal renal function was estimated to be 16.6 hours based on the population pharmacokinetic analysis.
Metformin
Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
For further information on Ertugliflozin /metformin, please see,
